Stening® Solid Stent MS03
Code MS03
Description
Stening® Solid Stent made out of silicone and intended for bronchial occlusion in the treatment of different bronchopleural pulmonary affections such as the bronchopleural fistula and persistent air loss in pneumothorax cases that cannot be treated with conventional surgery.
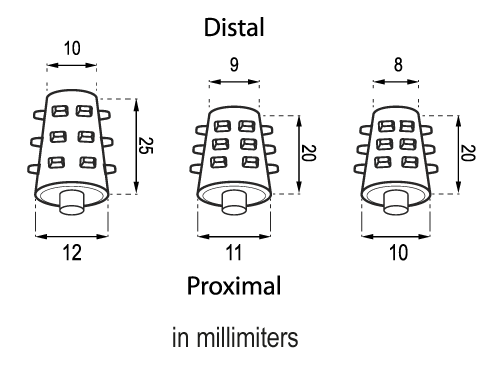
The Solid Stent is presented in three different sizes: MS810, MS911 and MS1012.
It is radiopaque (white or pale yellow).
Stening® provides detailed instructions for use with each device, including insertion and removal techniques, precautions, and postoperative care.

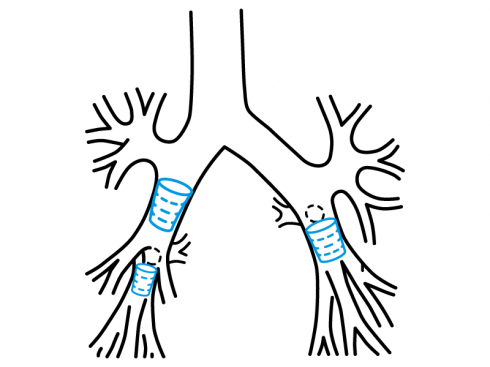
- Bronchopleural fistula.
- Suture failure in a stump after lobar pulmonary resection.
- Occluded bronchial treatment.
- Hemoptysis.

The sizes of the devices are subject to a variation of +/- 2%.
- Medical grade silicone
- Bevelled edges to prevent granulomas
- Spur system to prevent migration
- Removable
- Surface of maximum softness to avoid adherence of secretions
The Stening® Solid Stent is implanted with the help of a rigid bronchoscope and it will be required general anaesthesia.
Once the location spot has been determined and its size, which is established by comparison with the known diameter of the endoscopic instruments that are being used, one or a few solid stents with the right measures, that exceed the fistulous hole diameter or the bronchus diameter, will be chosen. This is done in order to adjust the stent to the bronchus or stump where it will be hosted.
The device has a flat appendage at its posterior end from where it will be taken with a rigid alligator forceps.
Then the stent-forceps assembly is introduced through the bronchoscope to lead it to its final destination in the bronchus or fistula that is desired to be occluded by introducing the Stening Solid Stent into the hole.
All the procedure described is carried out under direct vision using the optics for bronchoscopy.
The removal is done effecting inverse manoeuvres.
Due to its radiopaque properties, it can be identified in the radiographies.
- Maintain the moisture of secretions, whenever they appear, by taking nebulisations frequently with a warm isotonic saline solution.
- Perform a periodic check-up following your doctor’s advice.
Warning: This type of device may suffer an unexpected displacement. This will depend on the failed choice of stent caliber in relation to the bronchial or fistulous orifice where it has been placed, as well as the unpredictable individual reaction of the tissue that acts as support in the implant area. A device that has migrated may be removed by coughing or accidentally lodged in another bronchus in the same or adjacent lung, resulting in unwanted bronchial occlusion that may lead to severe ventilatory insufficiency and subsequent death.
A displacement of the occlusion device towards the pleural cavity in cases of post-pneumonectomy fistula is possible, and also its spontaneous elimination through an existing pleurocutaneous window.
An anomalous position may be suspected by the appearance of a sustained cough. The devices are radiopaque and their position can be identified on chest radiography. A displaced device must be removed by the interventional physician.
The product should not be reused because this can cause cross contamination.

